(New Version Available) Inverse Functions
New Version: https://youtu.be/q6y0ToEhT1E Define an inverse function. Determine if a function as an inverse function. Determine inverse functions. http://mathispower4u.wordpress.com/
From playlist Exponential and Logarithmic Expressions and Equations

Transcendental Functions 19 The Function a to the power x.mp4
The function a to the power x.
From playlist Transcendental Functions

Define an inverse function. Determine if a function as an inverse function. Determine inverse functions.
From playlist Determining Inverse Functions

AQA Core 3 2.04 Composite Functions
I introduce the idea of composite functions and explain what we mean by fg(x), gf(x), ff(x) or gg(x) via an example.
From playlist [OLD SPEC] TEACHING AQA CORE 3 (C3)

Lecture 04-Jack Simons Electronic Structure Theory- Linear combinations of atomic orbitals
The Hartree-Fock molecular orbitals; LCAO-MO expansion; Hartree-Fock equations in matrix form; one- and two-electron integrals; the iterative SCF process; scaling with basis set size; how virtual orbitals change with basis set; core, valence, polarization, and diffuse basis functions; Slat
From playlist U of Utah: Jack Simons' Electronic Structure Theory course

Lecture 03-Jack Simons Electronic Structure Theory- Dynamical pair correlations
Dynamical and essential electron correlation; polarized orbital pairs; dynamical correlation; configuration interaction; how important correlation is; reminder about cusps and introduction to explicitly correlated wave functions. (1)Jack Simons Electronic Structure Theory- Session 1- Bo
From playlist U of Utah: Jack Simons' Electronic Structure Theory course

Ex 1: Find the Inverse of a Function
This video provides two examples of how to determine the inverse function of a one-to-one function. A graph is used to verify the inverse function was found correctly. Library: http://mathispower4u.com Search: http://mathispower4u.wordpress.com
From playlist Determining Inverse Functions

Chem 131A. Lec 20. Quantum Principles: Hartree-Fock Calculations, Spin, and Slater Determinants
UCI Chem 131A Quantum Principles (Winter 2014) Lec 20. Quantum Principles -- Hartree-Fock Calculations, Spin, and Slater Determinants -- View the complete course: http://ocw.uci.edu/courses/chem_131a_quantum_principles.html Instructor: A.J. Shaka, Ph.D License: Creative Commons BY-NC-SA T
From playlist Chem 131A: Week 8

Lecture 02-Jack Simons Electronic Structure Theory- Hartree-Fock
Slater-Condon rules; the Hartree-Fock approximation; Coulomb and exchange interactions; Koopmans theorem, the meaning of orbital energies; Brillouin theorem; molecular orbitals are delocalized; reminder on the limitations of single determinant wave functions. (1)Jack Simons Electronic S
From playlist U of Utah: Jack Simons' Electronic Structure Theory course

Chem 131A. Lec 21. Quantum Principles: Bigger Atoms, Hund's Rules and the Aufbau Principle
UCI Chem 131A Quantum Principles (Winter 2014) Lec 21. Quantum Principles -- Bigger Atoms, Hund's Rules and the Aufbau Principle -- View the complete course: http://ocw.uci.edu/courses/chem_131a_quantum_principles.html Instructor: A.J. Shaka, Ph.D License: Creative Commons BY-NC-SA Terms
From playlist Chem 131A: Week 8
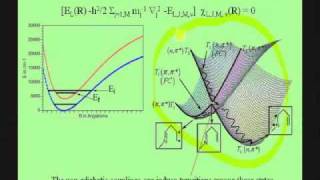
Lecture 01-Jack Simons Electronic Structure Theory- Born-Oppenheimer approximation
The Born-Oppenheimer approximation; non-adiabatic couplings; the electronic and vibration-rotation Schrodigner equations; atomic units; electronic cusps, electronic wave functions and energy surfaces; orbitals and spin-orbitals, Slater determinants; effects of antisymmetry; problems arisin
From playlist U of Utah: Jack Simons' Electronic Structure Theory course

How To Use Slater's Rule to Estimate The Effective Nuclear Charge
This chemistry video tutorial explains how to use Slater's Rule to estimate the effective nuclear charge of an electron in an atom. It could be a core electron or a valence electron. My Website: https://www.video-tutor.net Patreon Donations: https://www.patreon.com/MathScienceTutor Ama
From playlist New AP & General Chemistry Video Playlist

Ionisation Energy for the First 10 Elements (Physical Chemistry, Physics)
A consideration of how much an outer electron feels the effect of a nuclear charge after shielding effects of other electrons. quantum mechanical correction for exchange energy consideration due to exact electron configurations is then explored. #chemistry #pchem #atom #quantum #physicalc
From playlist Random Physical Chemistry

24. Molecular Orbital Theory I; Variational Principle and Matrix Mechanics
MIT 5.61 Physical Chemistry, Fall 2017 Instructor: Professor Robert Field View the complete course: https://ocw.mit.edu/5-61F17 YouTube Playlist: https://www.youtube.com/playlist?list=PLUl4u3cNGP62RsEHXe48Imi9-87FzQaJg This lecture begins the discussion on molecules and molecular orbital
From playlist MIT 5.61 Physical Chemistry, Fall 2017

Chem 131A. Lec 19. Quantum Principles: The Hydride Ion (Try #3!) The Orbital Philosophy
UCI Chem 131A Quantum Principles (Winter 2014) Lec 19. Quantum Principles -- The Hydride Ion (Try #3!) The Orbital Philosophy -- View the complete course: http://ocw.uci.edu/courses/chem_131a_quantum_principles.html Instructor: A.J. Shaka, Ph.D License: Creative Commons BY-NC-SA Terms of
From playlist Chem 131A: Week 8
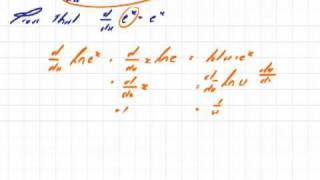
Transcendental Functions 9 Proof of the First Derivative of e to the x.mov
Proof of the first derivative of the function e to the power x.
From playlist Transcendental Functions